Dauwpunt in perslucht
Producten die regelmatig gebruikt worden in de perslucht industrie














High levels of moisture in breathing air makes it uncomfortable to breath and increases the risk of bacteria growing in the system which is a potential health risk. If humidity levels are too high in the compressed air, there is a risk of moisture condensing. This can cause airlines to become blocked, or cylinders to corrode.
The European Standard EN12021:2014 “Respiratory protective devices – Compressed Air for breathing apparatus” states that the pressure dew-point should be equal to or less than 11°C, or 5 degrees lower than the lowest application or storage temperature.
Compressed air used to power pneumatic tools and conveyers must be clean and dry to avoid contamination of products and corrosion of the equipment. The ISO Air Quality Standard ISO8573 is the international standard which sets out the requirements for compressed air purity and covers particulates and oil as well as moisture contamination.
For class 2 compressed air, a pressure dewpoint (PDP) of -40°C or better is required and no liquid water is allowed.
In medicinal applications compressed air is used for both breathing and as instrument air. As well as concerns for corrosion and breathing comfort, avoiding bacterial growth is a major concern.
There are a number of standards established and 2 common standards are : UK Health Technical Memorandum HTM02-01 for purity specification for medical gas, which determines moisture content ≤67 vpm (≤0.05mg/L, atmospheric dewpoint of -46°C) and European Pharmacopoeia 5.0 for medicinal air ≤67 ppm (*pressures greater than 10 bar and temperature higher than 5°C)
In dental air applications there are a number of recommended standards and one common standard is the UK NHS Technical Memorandum HTM2022 Supplement 1, which determines moisture content ≤1020vpm (≤0.78 mg/L, atmospheric dewpoint of - 20°C)
There are direct contact and indirect contact standards for food grade air within the food processing & food packaging industry.
The commonly referenced standards are as follows:
- Compressed air that comes in to direct contact with food or beverage products should meet or exceed the ISO 8573-1:2010
- Compressed Air Purity Class 2 pressure dew point ≤-40 °C.
- Compressed air that comes in to indirect contact with food or beverage products should meet or exceed the ISO 8573-1:2010
- Compressed Air Purity Class 4 pressure dew point ≤+3 °C
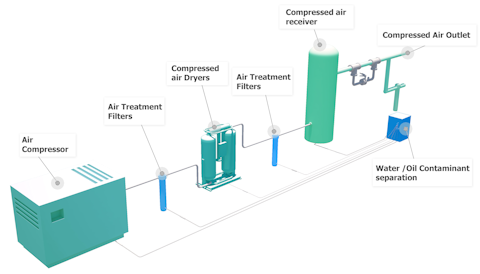

 Perslucht die wordt gebruikt om pneumatische gereedschappen en transportbanden aan te drijven, moet schoon en droog zijn om verontreiniging van producten en corrosie van de apparatuur te voorkomen. De ISO-luchtkwaliteitsnorm ISO8573-1 is de internationale standaard die de eisen voor de zuiverheid van de perslucht en de verontreiniging van deeltjes en olie alsmede vocht beschrijft.
Perslucht die wordt gebruikt om pneumatische gereedschappen en transportbanden aan te drijven, moet schoon en droog zijn om verontreiniging van producten en corrosie van de apparatuur te voorkomen. De ISO-luchtkwaliteitsnorm ISO8573-1 is de internationale standaard die de eisen voor de zuiverheid van de perslucht en de verontreiniging van deeltjes en olie alsmede vocht beschrijft.
 Hoge vochtgehaltes in ademlucht maakt het ademen ongemakkelijk, verhoogt het risico van bacteriegroei in het systeem en hiermee een potentieel risico voor de gezondheid. Als de luchtvochtigheid in de perslucht te hoog is ontstaat er het risico van vocht condensatie. Dit kan ertoe leiden dat luchtleidingen worden geblokkeerd of cilinders worden aangetast.
Hoge vochtgehaltes in ademlucht maakt het ademen ongemakkelijk, verhoogt het risico van bacteriegroei in het systeem en hiermee een potentieel risico voor de gezondheid. Als de luchtvochtigheid in de perslucht te hoog is ontstaat er het risico van vocht condensatie. Dit kan ertoe leiden dat luchtleidingen worden geblokkeerd of cilinders worden aangetast.
 In medische toepassingen wordt de perslucht gebruikt voor ademhaling en voor de aandrijving van instrumenten. Het vermijden van corrosie en bacteriële groei en het op peil houden van het ademhalingscomfort is een probleem, hierom is een nauwkeurige monitoring van het dauwpunt essentieel.
In medische toepassingen wordt de perslucht gebruikt voor ademhaling en voor de aandrijving van instrumenten. Het vermijden van corrosie en bacteriële groei en het op peil houden van het ademhalingscomfort is een probleem, hierom is een nauwkeurige monitoring van het dauwpunt essentieel.
 In toepassingen voor tandheelkundige lucht zijn er een aantal geadviseerde normen en één gebruikelijke norm is het UK NHS Technical memorandum HTM2022 supplement 1, dat een vochtgehalte voorschrijft van ≤ 1020 vpm (≤ 0,78 mg/L, atmosferisch dauwpunt van-20 °Cdp).
In toepassingen voor tandheelkundige lucht zijn er een aantal geadviseerde normen en één gebruikelijke norm is het UK NHS Technical memorandum HTM2022 supplement 1, dat een vochtgehalte voorschrijft van ≤ 1020 vpm (≤ 0,78 mg/L, atmosferisch dauwpunt van-20 °Cdp).
 Er zijn normen voor directe en indirecte contact met perslucht in de voedselverwerking & verpakkingsindustrie. Voor perslucht dat in direct contact komt met voedsel of drankproducten geldt de ISO 8573-1:2010 perslucht zuiverheid klasse 2 en is een dauwpunt voorgeschreven van ≤-40 °Cdp.
Er zijn normen voor directe en indirecte contact met perslucht in de voedselverwerking & verpakkingsindustrie. Voor perslucht dat in direct contact komt met voedsel of drankproducten geldt de ISO 8573-1:2010 perslucht zuiverheid klasse 2 en is een dauwpunt voorgeschreven van ≤-40 °Cdp.

